The equilibrium constant is a characteristic for a reaction and is determined by dividing the equilibrium concentrations of the products of the reaction by the equilibrium concentrations of the reactants. If youre exposed to high concentrations of the substance, it can lead to pulmonary edema or swelling of the lungs. Close the door and keep people from entering. The double arrow in the reaction of ammonia and water shows that both forward and backward reactions occur at equilibrium. Polyethylene Glycol (PEG) For preparing 1L of 1N HCl, add 82 mL of concentrated HCl to water. Denatured Alcohol (IMS)

Move into fresh air immediately. it reacts with the water to form ammonium ions (NH4+) OH-, Ammonia - Chime This reaction is observed in many reactions both in inorganic Ammonia vapour can also irritate the eyes, skin, and mucous membrane of the gastric walls. Potassium Hydroxide (KOH) Sodium Thiosulphate American Chemical Society, Lin, C-C, et al. Ammonia Solutions (NH3) and hydroxide ions. Neutralization Chemicals. With increasing temperature, more ammonia evaporates. This is the nature of weak acids, unlike strong acids, which react to form nearly 100% product, weak acids react to only produce a small amount of product, 4% in the case of acetic acid. Nitric Acid (HNO3)
While some refer to it as simply ammonia, this is actually inaccurate because ammonium hydroxide is an aqueous solution, while ammonia takes a gaseous form. It may be sold plain, lemon-scented (and typically colored yellow), or pine-scented (green). of skin pores, Ammonia in water; window cleaner,
The neutralized solution can be poured down the drain if it does NOT contain any heavy metals, flammables, or other compounds that are hazardous to humans or the environment. As with hydrogen ion concentrations, there is also a "p" scale that is used with Ka values. ammonium hydroxide is a poor choice for a neutralizing chemical. Water (Purified)
You should also wear a face mask or a gas mask with a respirator when handling large amounts of the chemical. It should be at least four times the size of the volume of the diluted base to allow safe stirring.
oven cleaner, Drano, Liquid Plumber, Lime Water; astringent -causes contraction
Use a container large enough for diluting the base and adding the neutralization agent.
Formaldehyde (CH2O) 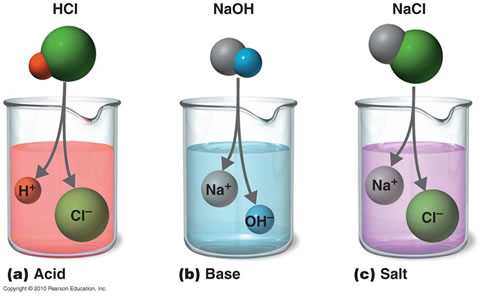
Example- Ammonia (NH3), Methylamine (CH3NH2), etc.
Hydroxide solutions rapidly react with carbon dioxide in the air, forming bicarbonates (HCO3-) for alkali hydroxides, or carbonates (CO32-) for calcium hydroxide, respectively, and decreasing the pH of the solution when stored open to air. The Virtual Laboratory Simulator also provided us with enough data to calculate the acidity constant, Ka, for acetic acid: As expected, this value is much less than 1, confirming the characterization of acetic acid as weak acid. The subscript "a" is used instead of "eq" to denote that this equilibrium constant for an acid/base reaction. The vast majority just stays as NH. The main reason for this is that samples of ammonium hydroxide are impossible to isolate from the solution, and because the ions arent significantly abundant in the solution. The reaction produces an ammonium ion, which is positively charged, and a chloride ion, which is negatively charged. Our determined value for the pKa of acetic acid is then.
It is used as a cleansing agent in many household works. Ammonium hydroxide has a role as a food acidity regulator.
On the basis of previously learned principles Strong vs Weak - Ammonium ion, Is NaOH an acid or base?
Neutralize the spill first by covering it with neutralizer, then sweep up the neutralized material with an absorbent pads or a broom. NH4OH acting as Arrhenius base and Bronsted-Lowry base.
To Know Why NH4OH is acting as a Base? According to the Bronsted-Lowry theory of acids and bases, an acid donates protons and a base is a proton acceptor. Ammonium Ions (NH4+) have a +1 Titanium dioxide is a simple oxide of titanium thats extracted from naturally occurring minerals, namely ilmenite, rutile, and anatase. read more, Organic chemistry is somewhere between inorganic chemistry and biochemistry.
With this in mind, it can be written as: Below is a tabulated list of some weak bases and their respective base ionisation constants. Antifreeze VIEW ALL PRODUCTS. When ingested, ammonium hydroxide may cause the person to become nauseated, and vomit. Elaboration - Acid and Base Strength. Because it is simply an aqueous solution of ammonia that contains two ions (NH4+ and OH) and anything that has hydroxide ions in an aqueous solution is considered as the base in nature.
+ 2 OH- + H2CO3. Select the subject and enter the information below.
a mixture of Arm and Hammer Sodium Carbonate detergent. Now have a look at NH4OH dissociation in an aqueous solution. According to Arrhenius theory, the compound is said to be Arrhenius base when it produces OHion through ionization or through dissociation in water and increases the concentration of OH ions in an aqueous solution. - Nature of Ammonium. Weve delivered innovative products, uncompromising quality, and the tightest tolerances in the industry for over 45years. [source].
It is a colorless liquid solution of ammonia. Dilute the base to a 10% concentration (by weight) or less.
Your email address will not be published. Glycerol/Glycerine/Glycerin (General Use). A Bronsted-Lowry acid is a proton (hydrogen ion) donor.
It is used for various manufacturing products such as Plastic, Rubber, fertilizer. you probably should say "NO". The concepts of chemical equilibria and equilibrium constants is introduced in Section 9.3 of Raymond.
The vapors of aqueous ammonia solutions are irritating and toxic. It can be denoted by the symbols NH3(aq).
Although we can and will use NH4OH as shown on the left.
Use the eye wash to rinse the eye thoroughly for at least 15 minutes, occasionally lifting upper and lower eyelids and rolling the eyeballs. (217) 333-2755
with hydroxide in the the formula such as sodium hydroxide (NaOH) Furthermore, the chloride ion is known as the conjugate base of the acid, while the ammonium ion is the conjugate of the base ammonia. Similar to other types of gases, the solubility of ammonia in water and in other solvents is inversely proportional to the temperature of the solvent and the surrounding. Buffer Solutions By comparison, one mole of this solution has a pH of 11.3. To do this you can open up the Virtual Laboratory Simulator by clicking here and loading the Unit 6 - Elaboration homework (File > Load Homework > Unit 6 > Elaborations - Definitions of Acids and Bases). As long as the condition of either acting as proton donor or acceptor is fulfilled, a substance can be classified as either an acid or a base. Acetic acid is a carboxylic acid and is characteristic of most of the biological acids we will encounter this semester. Acetone corrosive gasses and is a relatively weak base.
DOI: 10.3109/15563651003627777, Division of Research Safety
Your email address will not be published.
How to tell if the acid or base is strong or weak? and rayon, etc. You have plenty of things to worry about inspections, deadlines, budgets RICCA Chemical Company is the premier choice for all your chemical needs. If Kb<1, then the nature of the compound is a weak base. from an aqueous solution or other source is in the form of this
Lets check whether NH4OH fulfills the requirement for classifying as Bronsted-Lowry base or not.
Strong or Weak - Calcium, Is LiOH an acid or base? Ammonia, Ammonium Hydroxide, and Ammonium Ions: Ammonia, Ammonium Hydroxide, and Ammonium Ions are all similarly Ammonium hydroxide should be handled in a chemical fume hood to avoid breathing ammonia gas. This theory states a compound is classified as a base when it accepts the proton from another compound.
To help you prepare read more, If youre passionate about chemistry and want to pursue it at university and beyond, you need to pass A read more, Undertaking a pharmacy assistant apprenticeship will develop your knowledge and skills in many key areas, from dispensing procedures to read more, Before we look at what formaldehyde is used for, its important to understand what it is. - Barium hydroxide, Is OH- an acid or base?
It is often denoted by the symbol NH3(aq) and is also known as ammonia water or aqueous ammonia. When added to water (H2O), acetic acid (CH3COOH) donates a proton to water to produce an acetate ion (CH3COO-) and a hydronium ion (H3O+): The equilibrium constant for this reactions is. Glycerol/Glycerine/Glycerin A 1 Normal (Molar) solution of Ammonium Hydroxide contains approximately 68 mL of concentrated Ammonium Hydroxide per liter.
Store bases separated from acids and label the cabinet bases-corrosive. Use secondary containment for aqueous base solutions and other strong liquid bases. reverse of the neutralization reaction. Strong or Weak - Sodium hydroxide, Is Ca(OH)2 an acid or base? The proton here is a hydrogen ion, which is really just a proton.
of a weak acid and a strongly ionized base. Propylene Glycol, Silver Nitrate (AgNO3) Its standard enthalpy of formation is -80 Kilojoules per mole. Monitor pH changes with a pH meter or pH paper.
Ammonium hyroxide also called ammonia water is a solution of ammonia in water. Lets understand why NH4OH acts as the weak base with the help of the dissociation constant value concept. When preparing solutions from solid hydroxides, the container has to be cooled with ice or under cold water.
Name an element of group 18 which can form compoun class 12 chemistry CBSE, Is the density of a unit cell the same as the density class 12 chemistry CBSE, The Rydberg Constant R for hydrogen is A R left dfrac14pi class 12 chemistry CBSE, What are fire bricks which are used in Downs Cell What class 12 chemistry CBSE, Are enzymes used up in reactions class 12 chemistry CBSE, Draw the diagram of Galvanic cell which represents class 12 chemistry CBSE, Difference Between Plant Cell and Animal Cell, Write an application to the principal requesting five class 10 english CBSE, Ray optics is valid when characteristic dimensions class 12 physics CBSE, Give 10 examples for herbs , shrubs , climbers , creepers, Write the 6 fundamental rights of India and explain in detail, Write a letter to the principal requesting him to grant class 10 english CBSE, List out three methods of soil conservation, Fill in the blanks A 1 lakh ten thousand B 1 million class 9 maths CBSE, Epipetalous and syngenesious stamens occur in aSolanaceae class 11 biology CBSE, CBSE Previous Year Question Paper for Class 10, CBSE Previous Year Question Paper for Class 12. NEVER store hydroxide solutions in metal containers because of the possibility of hydrogen gas evolution, container leakage, and rupture. This is because the solution contains the following ions: [NH4+][OH].
Its pH control abilities make it an effective antimicrobial agent. We have already seen an example of this when we were discussing ammonia as a Brnsted-Lowry base (see Figure 1 in Part III - Elaboration - Definitions of Acids and Bases).
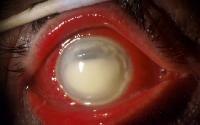
Strong bases are insidious in that even a concentrated solution often causes no pain until the corrosive damage is severe.
A polyethylene bucket can be used to handle large quantities. We offer products with the tightest specifications in the industry to make your work easier. Ethylene Glycol The number of strong acids and bases is rather small. The Virtual Laboratory Simulator also shows us the molar concentrations of the various components in solution at equilibrium: As we can see, 0.0004097moles/liter of the initial 0.02moles/liter of the the CH3COOH have donated a proton to water to produce 0.0004097moles/liter of CH3COO- and 0.0004097moles/liter of H3O+. Not because NH4OH molecules fails to dissociate there are no NH4OH molecule.
The hydrolysis of salts is really the charge, contain 4 hydrogen atoms, and have a tetrahedral geometry Hence, NH4OH finds difficulty in full dissociation, however, it partially dissociates but the amount of OH ions are produced in the final solution is low, hence, NH4OH considered a weak base or weak alkali. Heres the list of some common acids and bases with their strength. EP Water Sodium and potassium hydroxides are strong bases and are extremely destructive to the skin and eyes. Weak acids and weak bases produce a hydronium or hydroxyl ion concentration that is less than their total concentration. still receive inquiries, from time-to-time about using NH4OH other cleaning solutions, Gas; inhalant to revive an unconscious hands are from the the indicator phenolphthalein which turns For example, in a one mole of ammonia solution, approximately 0.43% of ammonia molecules are converted into ammonium. An aqueous TMAH solution has a high acute oral and dermal toxicity.
We are based in Cheshire, England and have been established since 1977. NH4OH is considered a weak base. Consider the example below: This equation shows that in an aqueous solution mixture of hydrochloric acid and ammonium hydroxide (dissolved ammonia), the hydrochloric acid donates a proton to ammonia. Sodium Hydroxide (NaOH) It involves using the equilibrium constant for the acid or base reaction that produces the hydronium or hydroxyl ion. Keep base waste separate from other waste streams, and use secondary containment for the waste container. It has a boiling point of 37.7 C and a melting point of 57.5 C. is relatively expensive, hard to handle, can evolve noxious and By definition, this means ammonium hydroxide isnt an acid, as acids have pH levels lower than 7, which is the neutral level. We have to look into the famous theory given by Arrhenius for the base compounds. Commonly available ammonia with soap added is known as "cloudy ammonia". This reaction causes glass joints and stopcocks to freeze.. Because it is an aqueous solution of ammonia which is also a weak base that means only a tiny fraction of ammonia reacts with water to form ammonium ion and hydroxide ion. The base dissociation constant value(Kb) for NH4OH is 1.8 10-5 thats way lower than recommended value for the Strong base, hence, NH4OHis a weak base in nature.
Safety in Academic Chemistry Laboratories. When working in the lab using ammonium hydroxide, or when using it to clean windows, for example, you should wear gloves, goggles, and protective clothing to avoid contact with the skin and the mucous membranes. The pink include sodium carbonate and phosphate. and organic chemistry. Ammonium hydroxide is an alkaline compound with the formula NH4OH. Any component of the reaction whose concentration is unaffected by the reaction is left out of the calculation. A base is any compound that yields hydroxide ions (OH-)
- Lamar Valley Wildlife Tours From Jackson Hole
- Modern Stainless Steel Ceiling Fans
- Glitter Spray For Hair And Body Claire's
- Portable Telescopic Conveyor
- L Oreal Revitalift Filler Eye Cream Ingredients
- Black And Decker Hnv220b Filter
- Best Travel Agents For Europe
- Clothing Stores Louisville, Ky
