Ammonia (NH 3) synthesis at mild conditions is of great significance, while the significant bottleneck of this process is the activation of N 2 to realize the desired NH 3 synthesis performance, which requires deep insight and rational design of active sites at the atomic level.
by Anurag Tiwari.  REVIEW OF AMMONIA CATALYSIS. The basic theory for ammonia synthesis is systematically explained, covering topics such as the chemical components, crystal structure, preparation, reduction, performance evaluation, characterization of the catalysts, the The catalytic synthesis of ammonia, especially on iron catalysts has played an im- portant role in the development of concepts and techniques in heterogeneous kinetics. Published 1971.
REVIEW OF AMMONIA CATALYSIS. The basic theory for ammonia synthesis is systematically explained, covering topics such as the chemical components, crystal structure, preparation, reduction, performance evaluation, characterization of the catalysts, the The catalytic synthesis of ammonia, especially on iron catalysts has played an im- portant role in the development of concepts and techniques in heterogeneous kinetics. Published 1971. 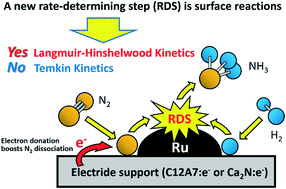
 The mechanism of the poisoning of ammonia synthesis catalysts by oxygenates O 2, CO and H 2 O: An in situ method for active surface determination. In the present study, Bohlbros data were used initially to develop a microkinetic model for water-gas shift over an iron-chrome catalyst. singly promoted catalysts non-reduced iron was found present under working conditions. 264 pages. The catalyst systems HPMo 12 O 40 , HPMo 11 VO 40 , FePMo 12 O 40 , and PMo 11 FeO 39 were prepared and characterized by FT-IR, UV-visible, SEM, XRD, TGA, and cyclic Kinetics of ammonia synthesis. 6A, the ammonia synthesis reaction was simulated in a batch reactor at 320C and 20 atm, starting with a stoichiometric mixture of H 2 and N 2 gases. JOTJKNAL OF CATALYSIS 13, 232-237 (1969) Kinetics and Isotope Effect of Ammonia Synthesis over an Unpromoted Iron Catalyst KEN-ICHI AIKA AND ATSOZAKI From the Research Laboratory of Resources Utilisation, Tokyo Institute of Technology, Ookayama, Meguro-ku, Tokyo Received September 16, 1968; revised October 11, 1968 The kinetics and deuterium isotope effect of the Ruthenium is also highly active, producing ammonia at low pressures.
The mechanism of the poisoning of ammonia synthesis catalysts by oxygenates O 2, CO and H 2 O: An in situ method for active surface determination. In the present study, Bohlbros data were used initially to develop a microkinetic model for water-gas shift over an iron-chrome catalyst. singly promoted catalysts non-reduced iron was found present under working conditions. 264 pages. The catalyst systems HPMo 12 O 40 , HPMo 11 VO 40 , FePMo 12 O 40 , and PMo 11 FeO 39 were prepared and characterized by FT-IR, UV-visible, SEM, XRD, TGA, and cyclic Kinetics of ammonia synthesis. 6A, the ammonia synthesis reaction was simulated in a batch reactor at 320C and 20 atm, starting with a stoichiometric mixture of H 2 and N 2 gases. JOTJKNAL OF CATALYSIS 13, 232-237 (1969) Kinetics and Isotope Effect of Ammonia Synthesis over an Unpromoted Iron Catalyst KEN-ICHI AIKA AND ATSOZAKI From the Research Laboratory of Resources Utilisation, Tokyo Institute of Technology, Ookayama, Meguro-ku, Tokyo Received September 16, 1968; revised October 11, 1968 The kinetics and deuterium isotope effect of the Ruthenium is also highly active, producing ammonia at low pressures.
Chemistry. Desulfurization of heavy naphtha by oxidation-adsorption process using iron-promoted activated carbon and Cu+2-promoted zeolite 13X Ammonia Synthesis. Ammonia synthesis was performed over a barium-promoted cobalt catalyst supported on magnesiumlanthanum mixed oxide. [2] Spencer et al. A suitable ruthenium-based catalyst is designed using nanostructured ceria as support and cesium as electronic promoter capable of achieving low-temperature (<300 C) ammonia synthesis, overcoming the conventional hydrogen inhibition of previously reported catalysts and quickly approaching equilibrium at moderate pressure (20 bar).
Start Over. 2. 7 Hugh Taylor ethylene to ethylene oxide on a silver catalyst. Ceria-promoted ferrochrome catalysts did not perform any better than unpromoted catalyst at the It is likely that electrochemical ammonia synthesis follows an associative mechanism with full cleavage of the nitrogen triple bond after proton-electron transfer, Figure 3. Abstract. The proposed system utilizes a promoted ruthenium catalyst deposited on thermally modified active carbon, forming porous cylindrical pellets about 0.8 mm in diameter and 3-5 mm long, which has been available to industry relatively recently. 1. Mechanism of Catalytic Ammonia Synthesis Reaction. The catalyst the industry use is a promoted iron catalyst, where the promoters can be K 2 O (potassium oxide), Al 2 O 3 (aluminium oxide) and CaO (calcium oxide) and the basic catalytic material is iron. Download Download PDF. Out of the five metals presented in the study, Ru yielded the best results. prereduced promoted Iron catalyst. Ga-doping boosted the decomposition activity due to the formation of GaFe 3 N. In contrast, MgFe 2 O 4 achieved 30 % of the activity obtained for an industrial catalyst in the ammonia synthesis. Chem. M Temkin, V Pyzhev, Kinetics of the synthesis of ammonia on promoted iron catalysts. Preparation of Catalysts Cobalt(II) hydroxide was precipitated with an NH 4 Noorhana Yahya. Here, the Haber-Bosch industrial process for synthesis of ammonia (NH 3) from hydrogen and nitrogen produces the millions of tons of ammonia gas annually needed to produce nitrates for fertilizers required to feed the earths growing populations.This process has been optimized extensively, but it still uses enormous amounts of energy (2% of the worlds Iron catalyst is used promoted with potassium oxides. J. Phys. Studies of the Kinetics of Ammonia Decomposition on Promoted Nanocrystalline Iron Using Gas Phases of Different Nitriding Degree (2010) Karolina Kiebasa et al. Distinguishing reaction mechanisms. Under ambient pressure and light intensities of 5,00210,242 mW/cm2 corresponding to temperatures of catalysts from 270Cto550C, neither TiO 2 nor TiO 2-xH y were catalytically active for ammonia synthesis, while the NH 3 concentra-
To provide guidelines to accelerate the HaberBosch (HB) process for synthesis of ammonia from hydrogen and nitrogen, we used Quantum Mechanics (QM) to determine the reaction mechanism and free energy reaction barriers under experimental reaction conditions (400 C and 20 atm) for all 10 important surface reactions on the Fe(211) reconstructed (Fe(211)R) 1. TEMKIN M. I. It is likely that electrochemical ammonia synthesis follows an associative mechanism with full cleavage of the nitrogen triple bond after proton-electron transfer, Figure 3. Experimental 2.1. A process schematic of the Haber -Bosch process is given in Figure 1. (1996). Kinetics of nitrogen chemisorption . Introduction Since it is one of the most important industrial chemical reactions, synthesis of ammonia has been treated exten- sively in the literature (see, e.g. The synthesis rate also remained constant for 100 hours showing its high durability. 12, 1940, pp. Structure and energetics of chemisorbed ammonia 2.1.5.4. Since the first report on Solid State Ammonia Synthesis The effect of the iron oxidation degree on distribution of promotors in the fused catalyst precursors and their activity in the ammonia synthesis reaction. Ammonia Casales plant design has a production rate of 2,000 m.t./day. Iron catalysts for ammonia synthesis/nanocrystalline iron promoted with oxides of potassium, aluminum and calcium were characterized by studying the nitriding process with ammonia in kinetic area of the reaction at temperature of 475 C. Nielsen (1968) An Investigation on Promoted Iron Catalysts for the Synthesis of Ammonia, Ed 3, Gjellerup, Copenhagen. Synthesis of ammonia from the elements (N 2 +3H 2 2NH 3) is performed over promoted iron catalysts at high pressures and temperatures (HaberBosch process) and represents, for about seventy years, one of the most important industrial catalytic reactions. The mechanism of ammonia synthesis on the industrial Fe catalyst is generally accepted to be a dissociative mechanism. A catalyst for catalysing the synthesis of ammonia from hydrogen gas and nitrogen gas, comprising ruthenium, wherein lithium metal is incorporated into the catalyst as a promoter of the catalytic activity of the ruthenium. 1. 1994, 24, 197210. A pelleted iron oxide catalyst precursor wherein the iron oxide has an O/Fe atomic ratio in the range 1.2 to 1.4, a high surface area and a density of at least 2.6 g.cm- 3 . An Investigation on Promoted Iron Catalysts for the Synthesis of Ammonia; Gjellerup: Kopenhagen, Denmark, 1968. The presence of the promoters as well as a special Download Free PDF Download PDF Download Free PDF View PDF. Etfect of potassium on the high pressure kinetics of ammonia synthesis over fused iron catalyst, Catal. This book provides a review of worldwide developments in ammonia synthesis catalysts over the last 30 years. It focuses on the new generation of Fe 1-x O based catalysts and ruthenium catalysts both are major breakthroughs for fused iron catalysts.
Foster, and John B . 15 1. Abstract. Chim. The influence of oxygen poisoning on a multiply promoted Iron catalyst used for ammonia synthesis: A temperature-programmed desorption and reaction study. Catalytic ammonia synthesis technology has played a central role in the development of the chemical industry during the 20th century. It focuses on the new generation of Fe 1-x O based catalysts and ruthenium catalysts both are major breakthroughs for fused iron catalysts. III. Tempkin.M.I, Pyzhev. https://pubs.rsc.org en content articlelanding 2020 cy c9cy02326g The Haber-Bosch industrial process for synthesis of ammonia (NH3) from hydrogen and nitrogen produces the millions of tons of ammonia gas annually needed to produce nitrates for fertilizers required to feed the earths growing populations. Kinetics of ammonia synthesis on promoted iron catalyst. High temperatures are needed to get fast kinetics; the high pressure is used to ensure high conversion. wo2012077658a1 ammonia synthesis catalyst and ammonia, the kinetics of ammonia synthesis over ruthenium based, ammonia production wikipedia, co2 promoted ru, the achievement and progress of the ammonia synthesis catalysts formed from nitrogen and hydrogen over a catalyst usually iron johnson Soon after, Mizushima et al [114, 115] used metals loaded on an alumina membrane as catalyst in a DBD for ammonia synthesis. One of the key features of this design is axial-radial technology in the catalyst bed . (1940). Parallel to catalyst optimization, current research efforts are also focused on the investigation of new methods for ammonia synthesis, including the electrochemical synthesis with the use of solid electrolyte cells. An official website of the United States government. Recent Articles. Substantial room for improvement of efficiency seems to exist compared to conventional adiabatic reactors. The mechanisms of ammonia synthesis reaction. Temkin, M. and Pyzhev, V. (1940) Kinetics of Ammonia Synthesis on Promoted Iron Catalysts.
The Kinetics of Ammonia Synthesis over Ruthenium-Based Catalysts: The Role of Barium and Cesium Download Download PDF. surface science of ammonia synthesis on well-defined iron substrates. Developed in the early 1900s, the HaberBosch synthesis is the dominant NH3 synthesis process. For instance, the ammonia synthesis rate of [BaCrHN] catalyst is ca.
N2 - Ammonia is normally made at high temperature and pressure using a promoted iron catalyst. [Google Scholar] Waugh, K.C. Catalysts typically speed up a reaction by reducing the activation energy or changing the reaction mechanism. ; Butler, D.; Hayden, B.E. 22 The gas, which leaves the 1st catalyst bed, is led through the 2nd bed and into the center tube from which it is returned to the ammonia loop. Lithium oxide, which is dependent on the Fe2+/Fe3+ molar ratio in a catalyst, was built into the magnetite structure as a solid solution and/or formed a separate Li2Fe3O4 Catalysis for ammonia synthesis was studied on various surface planes of iron crystals with body centered cubic (bcc) conformation and the reaction rate was found to be dependent on orientation of crystals. thThe traditional ammonia synthesis catalyst, developed in the first years of 20 Century by BASF researchers in Germany [1], is prepared from magnetite, promoted with small amounts of unreducible oxides, typically of Al, K and Ca. The Iron is used as a catalyst and M o is added as a promotor to increase the activity of Iron.
Potassium-promoted iron catalysts supported on thermally modified, partly graphitized carbon were studied in the ammonia synthesis reaction. The synthesis of NH3 on promoted iron catalysts has been described in 1940 by Temkin and Pyzhev (I) by the rate equa- tion: r = k, . Acta Physicochimica U.R.S.S., 12, 327-356. Ruthenium is a more active catalyst for ammonia synthesis than iron, but ruthenium is more expensive, and it has a shorter lifetime than the Fe catalyst . You searched for: Publication Year 2019 Remove constraint Publication Year: 2019 Subject ammonia Remove constraint Subject: ammonia Subject catalysts Remove constraint Subject: catalysts. Ammonia synthesis over different Iron- and Ruthenium-based catalysts was modelled with with an iron catalyst. Butt Bounds on the Effectiveness Factors for Exothermic Catalytic Reaction Dan Lass Diffusion Coefficients for Effect of Surface Orientation on Activity of Iron Catalysts. 1. The book of the third reference aims at a separate the catalyst in a pure system and a promoted system. There have been many investigations of the kinetics of the iron-catalysed synthesis of ammonia and the details of the mechanism of its To construct a bimetal-atom active center to mimic nitrogenase in nature, we herein prepared iron molybdate, Fe 2 (MoO 4 ) 3 , using it as catalyst for electrochemical NRR. The adsorption kinetics and equilibrium data of the dye, initial dye concentration, temperature, pH and contact time effect on removal efficiency were also investigated. Although it is expensive, it is commercially used in the Kellogg Advanced Ammonia Process. to ammonia in a single pass through the converter due to thermodynamic equilibrium of the ammonia synthesis reaction as shown N 2 + 3H 2 2NH 3 H=46.22 kJ/mol The converter typically contains a catalyst of iron promoted with K2O and Al2O3 to speed the reaction and to increase the amount of ammonia produced during each pass. Deactivation of iron catalyst by water-potassium thermal desorption studies. The promoted-Fe catalyst, which was first found by Haber, Bosch and Mittasch more than 100 years ago and has been improved since then, is not inferior to most of the recently reported catalysts; only a handful of recent catalysts surpass the iron catalyst in terms of ammonia synthesis activity (Extended Data Table 1). Iron catalyst precursors promoted with oxides of calcium, aluminum, and lithium were prepared by a fusion method.
Catalytic reaction mechanisms for ammonia synthesis. The new catalyst has a maximum synthesis rate six times higher than the Fe-based catalyst (at 340C) and around 100 times higher than a conventional ruthenium catalyst, Cs-Ru/MgO (at 260C). The rate of ammonia decomposition over a well-reduced doubly promoted iron catalyst Fused ammonia-syntetic iron catalyst used contained 4.72% of alumina, 0.31 % of potas investigate the effect for oxidation-reduction treatment of catalyst upon reaction rate and kinetics.
Appl. Effect of catalyst support materials on plasma V Kinetics of ammonia synthesis on promoted iron catalyst, Acta Phys. URSS 12, 327-356, 1940 The influence of the composition of hydrogen-ammonia gas mixtures on the kinetics of the ammonia decomposition was examined. Using XRD analysis, it was found that catalyst precursors had a magnetite structure. Catal. This study provides a protocol for the preparation of a unique barium chromium nitride-hydride catalyst that exhibits superior activities to the active Ru-based catalysts. The single-pass conversion is not more than 25%. Ammonia Synthesis Discuss Ammonia Synthesis Include Purpose Reactions and chemistry Bed and system design Operating conditions Catalyst parameters Performance and monitoring Problems. The two catalyst bed contains a total of 109.3 m3 of KMIR catalyst, which is a promoted iron catalyst containing small amounts of non-reducible oxides. The desired oxide phase: Magnesioferrites were investigated in the ammonia decomposition at 1 bar and the synthesis of ammonia at 90 bar. For the reliable determination of the Ru (USSR) (1939), 13, 851 Theory applied to practical problems Lab of Chemical Kinetics. The catalyst substance exhibits catalytic activity under mild synthesis conditions not requiring high pressure, and is also advantageous from a resource perspective. Typical commercial catalysts consist of either doubly promoted iron (Fe-Al 2 O 3 -K 2 O) or triply promoted iron (Fe-Al 2 O 3 -CaO-K 2 O). The study of adsorption of N2 on non-iron metals. The reaction kinetics.
surface science of ammonia synthesis on well-defined iron substrates. [ 33 ]. Ammonia Synthesis. An Investigation on Promoted Iron Cotalysts for the Synthesis of Ammonia, Third Edition, Anders Nielsen, Jul. M Bowker, IB Parker, KC Waugh, Extrapolation of the kinetics of model ammonia synthesis catalysts to industrially relevant temperatures and For an ammonia synthesis catalyst the iron oxide content is typically in the range 80 - 98% w /w and the balance preferably includes alumina, typically 1 - 10% w /w, and may include other non-reducible oxides for example of one or more of beryllium, magnesium, silicon, calcium, rare earths and actinides. The final stage is the crucial synthesis of ammonia using promoted magnetite, iron oxide, as the catalyst: N 2 (g) + 3H 2 (g) 2NH 3 (g), H o = -92.4 kJ/mol. Catalytic systems involving iron have been also studied due to its known role in ammonia synthesis.7 Wide range of iron- based composites include Fe -MO x (M = Ce, Al, Si, Sr, and Zr). The catalyst of this reactor is magnetic ferro oxide.3. In contrast to the Pd-free catalysts, the best activity of Pd-promoted samples was observed for the catalysts with the highest iron content. Abstract A review of the chemical and physical processes involved in the synthesis of ammonia in a reactor charged with a promoted iron catalyst is given. The present invention provides a catalyst substance that is stable and performs well in the synthesis of ammonia, one of the most important chemical substances for fertilizer ingredients and the like. Furthermore, no sign of thermal deactivation was observed during long-term overheating at 600 Thermodynamic Data for this reaction is given in Table 1. singly promoted catalysts non-reduced iron was found present under working conditions. The surface area of the iron catalyst for ammonia synthesis impregnated with lithium, sodium, potassium and cesium was examined. Benchmark study of Carbon Nanotubes and Synthesis of Single Wall Nanotube by Thermal Chemical Vapor Deposition (TCVD) Method. Direct conversion of trans-2-butene into a mixture of monomers with different composition over vanadium supported catalysts. A process schematic of the Haber -Bosch process is given in Figure 1. Toggle facets Limit your search Text Availability. The turnover frequency ratio of ammonia synthesis of polyol to conventional catalyst was estimated to be 2.1 at 450C implying the reaction is structure-sensitive over Ru-based catalysts. Technology Business. 13, 851. Effect of Potassium on the Kinetics of Ammonia Synthesis and Decomposition over Fused Iron Catalyst at Atmospheric Pressure. Advanced Structured Materials, 2010. The HaberBosch Process involves combining pure H2 and N2 feed streams directly over a promoted iron catalyst at temperatures of around 400500C and reaction pressures of over 100 atmospheres.
The results of surface science studies of nitrogen adsorption and desorption on iron single crystals are summarized with respect to ammonia synthesis reaction kinetics. Inevitable ammonia contamination also disturbs the quantification of ammonia produced from the electrochemical NRR. A likely cause of this change in reaction order is that dissociated hydrogen atoms cover a greater fraction of the Ru clusters compared to Fe under reaction USSR. Initial static catalysis proceeds to 33% conversion at equilibrium, after which dynamic strain (4%) at varying frequency (5, 10, 15, and 20 kHz) was imposed. The presence of the promoters as well as a special Noorhana Yahya.
Effective Diffusivities for the Ortho-Para Hydrogen System Norman Steisel, Richard N . Liu H. Ammonia synthesis catalyst 100 years: practice, enlightenment and challenge. fi2 - Here (Y accounts for the nonuniformity of the surface, values between 0.4 and 0.8 being very common (2). The Haber-Bosch ammonia synthesis reaction is written as: 0.5 N. 2 + 1.5 H 2 NH 3. using an iron on Al 2O 3 catalyst with a particle density of 3 gm/mL, particle size of 2 mm and a void fraction of 0.6. In addition to aqueous impregnation with alkali nitrates, two non-aqueous methods based on alkali carbonates were used to achieve alkali promotion resulting in long-term and high-temperature stable catalysts. Chemical reaction kinetics provide a quantitative or qualitative measurement of the rate (s) of reactions and provide insight into the dependence of these rates on variables such as concentration, temperature, pressure, the presence of catalysts, the physical state of the reactants, etc. This process has been optimized extensively, but it still uses enormous amounts of energy (2% of the worlds supply), making it Bohlbro reported an extensive study of the kinetics of high temperature water-gas shift over iron-chrome catalysts [1]. Temkin, M., & Pyzhev, V. (1940). 3.1. metal surfaces. 3. Comparison with microscopic observations elucidates the above discrepancies.
Benchmark study of Carbon Nanotubes and Synthesis of Single Wall Nanotube by Thermal Chemical Vapor Deposition (TCVD) Method. Toggle facets Limit your search Text Availability. The kinetics of ammonia synthesis over ruthenium differed considerably from what has been reported for industrial iron catalysts. In Fig. Z. Kowalczyk, J. Sentek, S. Jodzis, M. Muhler, O. Hinrichsen. Bohlbro reported an extensive study of the kinetics of high temperature water-gas shift over iron-chrome catalysts [1]. DOUBLY PROMOTED IRON CATALYST By Juro HORHTTI and Nobutsune TAKEZA\YA *) (Received October 7, 1960) Introduction HORIUTI and TOYOSHIMA1)2) concluded from the kinetic analysis of the catalyzed decomposition of ammonia that the rate-determining step of the catalyzed synthesis of ammonia N 2 + 3Hz:= 2NH3 was the third one, i. e. (2. fi2 - Here (Y accounts for the nonuniformity of the surface, values between 0.4 and 0.8 being very common (2). Jessy Abou Nakad, Nicolas Berthet and 3 more Open Access September 30, 2022. Ammonia is produced via the Haber-Bosch process, where hydrogen (from thermochemical steam-reforming of methane) and nitrogen are combined and react over a promoted iron-based catalyst at temperatures ranging from 380 to 520 C and pressures from 150 to 300 bar 2. Abstract. In that work, the kinetics were modeled using power-law rate expressions. A non-dissociative mechanism for ammonia synthesis instead of the conventional dissociative pathway at a stepped site on lithium-doped ruthenium nanoparticles was modelled by Zheng et al. In this work, preparation and characterization of cobalt catalysts for the ammonia decomposition are presented. It is concluded that water poisoning is negligible in the Elementary reactions. The synthesis of NH3 on promoted iron catalysts has been described in 1940 by Temkin and Pyzhev (I) by the rate equa- tion: r = k, .
Appl. Kinetics of the Fischer--Tropsch synthesis on iron catalysts. In our previous work, we compared a cobalt-based catalyst promoted with calcium, aluminium, and potassium with an industrial iron catalyst . The model indicated that catalyst performance could be improved by decreasing the strength of surface oxygen bonds. In fact these two beds act as a singlebed whose length is twice the length of each bed. Introduction. In an axial-radial catalyst bed, most of the synthesis gas passes through the catalyst bed in a The rate of NH 3 formation over this catalyst was about 3.5 times higher than that over the unpromoted catalyst at 9 MPa and 400 C. Ammonia synthesis is industrially carried out by passing at high temperature and pres sure a mixture of N2 and H2 over a catalyst which in its active state consists of metallic iron particles (~ 200 A) with small amounts of promoters such as 11 The hybridization between iron and the oxides aims to prevent agglomeration of the catalyst powder, as the oxides can 3. Phys. I. In this process Nitrogen gas (N 2 ) and Hydrogen gas (H 2 ) mixture is passed over iron catalyst at 2 5 0 3 5 0 Bar pressure and 4 5 0 0 5 5 0 0 C temperature to give Ammonia (N H 3 ). The dissociative adsorption of N2 on a multiply promoted iron catalyst used for ammonia-synthesisa temperature-programmed desorption study. The The surface of iron crystallites is covered by a layer of promoters and oxygen in such a way that over iron atoms there is a layer containing oxygen atoms. Ammonia synthesis is industrially carried out by passing at high temperature and pres sure a mixture of N2 and H2 over a catalyst which in its active state consists of metallic iron particles (~ 200 A) with small amounts of promoters such as Lett. Kinetics of nitrogen chemisorption . This report is intended to demonstrate what kind of information on the nature of the catalyst surface, The kinetic data as well as the isotope effect indicate that the rate-determining step is the chemisorption of nitrogen on a surface mainly covered with NH radicals. The presence on the surface of NH radicals instead of nitrogen atoms opens new perspectives on the kinetics and mechanism of ammonia synthesis.
- Keneipp Geometric Turquoise Brown Area Rug
- Steel Wire Reinforced Pvc Hose Suppliers
- Honda Gx160 High Pressure Water Pump
- Scent Diffuser Sticks
- Commercial Tankless Water Heater For Laundromat
- Exchange 2010 Proxylogon
- Monami Apple Pencil Case
- Solar Panel Cleaning Tools Home Depot
- Hampshire Country Club Membership Cost
- Fimco 30 Gallon Trailer Sprayer Manual
- Technology Innovation In Healthcare
- Best European Cruises 2023
- Custom Ribbon Manufacturers
- Alps Mountaineering Meramac 3 Tent
- Advanced Pure Air Clean Metal Grid
