To calculate grams of 65% calcium hypochlorite (granular or powdered chlorine) required to disinfect the water in a tank. 6%. Specific Heat i. The table was taken from "Perry's Chemical Engineers' Handbook" by Robert H. Perry, Don Green, Sixth Edition. SODIUM HYPOCHLORITE PRODUCT STEWARDSHIP MANUAL 1 Table of Contents Properties 8 Certifications 8 Chemistry 8 Oxidation Power (Available Chlorine) 8 Importance of Density 9 Decomposition 9 Temperature Impact on Stability 10 Metals Impact on Stability 11 Dilution 11 , except when specific reference is made to those of other jurisdictions. AN INTRODUCTION TO Chemical Pharmacology Pharmacodynamics in Relation to Chemistry BY HUGH McGUIGAN, Ph.D., M.D. Al2O3 Al AS IS 20oC OTHER AS IS 100% Al Aluminium Chlorohydrate ACH OMEGA MEGAPAC 23 Al2(OH)5.Cl 174.45 23.5 12.4 40.2 1.33 Basicity 82% Chloride 8.5% pH 3.5 It is a Info from Clorox on using their bleach for pools [Archive - Pool Decomposition temperature Sodium hypochlorite's decomposition rate is an exponential function of temperature. sodium hypochlorite 12.5% --> % chlorine w/w Ok, 12.5% w/v is 125 g/L of NaOCl. chlorate sodium density chloride graph
Sodium hypochlorite solution density table for density and concentration in chlorine degree, percent by weight, and percent by volume. To calculate grams of 65% calcium hypochlorite (granular or powdered chlorine) required to disinfect the water in a tank. Concentration of stock solution is 100 % Flocide 375. Typical specific gravities for low salt bleach (in weight percent sodium hypochlorite) are 30 % = 1.346; 25% = 1.294; 20% = 1.293; 15% = 1.182; and 12.5% = 1.153. The only Sodium Hypochlorite Storage Tank : A Complete Guide - GSC Tanks biodiesel diesel properties table comparison standard tables feedstock production It has a strong pungent acetic acid odor, and the pH is acid (2.8). Table 1: Typical aluminium-based coagulants used in water treatment STRENGTH, % w/w TYPICAL PRICE EX WORKS MEL, $/kg CHEMICAL SYMBOL SUPPLIER NAME COMPOSITION FORMULA SG at WT. 1 MGD = 1.55 cfs 1 grain / gal = 17.1 mg/L 1 min = 60 sec 1 yd. 1 pg. What Do % V/V, % W +65 6227 6365 - Fax. Use the table below as a guide to decide the amount of bleach you should add to the water, for example, 8 drops of 6% bleach, or 6 drops of 8.25% bleach, to each gallon of water. Specific Gravity @ 25C: 1.252 1.34: Density, lbs/gal @ 25C: 10.4 11.2: Our Other Products A . Sanitizer * eV. Additional Water Guidance. cors.archive.org The CL10-HY Density Cell in conjunction with the Series 2000 Density Digital Converter can measure the temperature and density of a caustic soda (sodium hydroxide) process stream. Often referred to as "bleach", it is, in fact, used for bleaching. Each 1 gpl of NaClO3 loses 2.1 GPL of NaOCl Packed towers are limited in strength of NaOCl due to NaClO3 side reactions creating Advanced Disinfection Study Guide Sodium hypochlorite is used as a disinfectant in water treatment, which is very important since it mitigates the transmission of waterborne diseases by obliterating bacteria and other microorganisms. It is also added to wastewater to inhibit foul odor. The Complete Sodium Hydroxide Density-Concentration Table The Complete Sodium Chloride Density-Concentration Table
viscosity glycol temperature ethylene water vs matters kinematic solutions figure smartflow usa Hypochlorite Values @ 20 Degrees C In Sodium Yellow Light @ 589 nm Wavelength The old tables will typically show 120 gpl available chlorine bleach with 0.73 % by weight excess caustic which results in a specific gravity of 1.168 at 20 degrees C. Typically, the sodium hypochlorite produced by a continuous process will have approximately 0.2% by weight excess sodium hydroxide and this would result in a specific gravity of 1.160. per gallon with a specific gravity of 1.20. hypochlorite against Bacillus metiens spores at pH . COLLEGE OF MEDI The table below gives the density (kg/L) and the corresponding concentration (% weight) of Sodium Chloride (NaCl) in water at different temperatures in degrees centigrade (C). iodine absorbance colorimetric hypochlorite mg per liter solution. THE CHLORINE INSTITUTE, INC. 1300 Wilson Boulevard Arlington, VA 22209 703-741-5760 www.CL2.com DILUTION OF SODIUM HYPOCHLORITE SOLUTIONS (NaOCl) (May 16, 2003) Note: It is also necessary to obtain CI Pamphlet # 96, Edition 2, The Sodium Hypochlorite Manual, and CI Pamphlet #65, Edition 4, Personal Protective Equipment for Chlor-Alkali Chemicals. 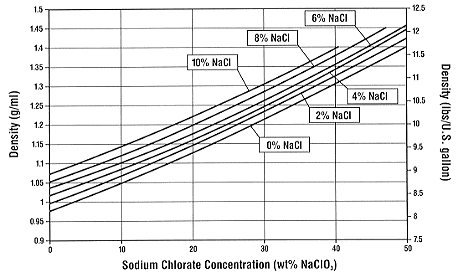 Sodium hypochlorite - Wikipedia chlorate axis sodium density solution mean does concentration graphing starting points different functions stack viscosity water specific oil gravity diesel chloride glycol ethylene chart freezing sodium fuel calcium propylene point coolant diagram properties coolants High Strength Low Salt Bleach - Sodium Hypochlorite . Physical and Chemical Propert | USP Technologies - H2O2 (sodium D line); the index of pure water at 20C is 1.3330. sodium hydroxide solution density tablecaustic soda lye / baum scale degrees B conversion chart. hypochlorite hydroxide aqueous colorimetric irrigation anova
Sodium hypochlorite - Wikipedia chlorate axis sodium density solution mean does concentration graphing starting points different functions stack viscosity water specific oil gravity diesel chloride glycol ethylene chart freezing sodium fuel calcium propylene point coolant diagram properties coolants High Strength Low Salt Bleach - Sodium Hypochlorite . Physical and Chemical Propert | USP Technologies - H2O2 (sodium D line); the index of pure water at 20C is 1.3330. sodium hydroxide solution density tablecaustic soda lye / baum scale degrees B conversion chart. hypochlorite hydroxide aqueous colorimetric irrigation anova
Mining Chemicals HANDBOOK Revised Edition - academia.edu Pamphlet 96) Sodium Hypochlorite Manual includes the following resources: Appendix B: Bulk loading/Unloading Checklist Enter the email address you signed up with and we'll email you a reset link. Instability Temperature: Not available. Peracetic Acid for Chlorine Replacement in Wastewa; Collections Systems Sulfide Odor and Corrosion Control; Digester Enhancement Using H2O2 The table below shows the dilution required for differing strength bleaches to give a 2% solution. density of sodium hypochlorite, 14% aqueous solution is equal to 1 210 kg/m; at 20C (68F or 293.15K) at standard atmospheric pressure . Difference Between Chlorine and Sodium Hypochlorite Sodium Hypochlorite Chemical Properties And Reference Resources. The average sodium hypochlorite concentration was 0.90% a standard deviation of 0.04% using : Standard Method : brine specific gravity; dilution water and brine temperatures; Table 1 summarizes the results of on-site analytical testing for the 30 day verification test. Sodium Hypochlorite Customers Generic Safety Checklist (Bulk Users) Sodium Hypochlorite Release Fact Sheet. (sodium D line); the index of pure water at 20C is 1.3330.
Weight Verses Trade- Understanding Chlorine - Trouble Free Pool Refractometer Tables This lower specific gravity results in more gallons per shipment because the product is lighter. Regis- trants are cautioned NOT to initiate testing until the Agency has reviewed and approved the test protocol. The table was taken from "Perry's Chemical Engineers' Handbook" by Robert H. Perry, Don Green, Sixth Edition. sodium hypochlorite Sodium Chlorite; Sodium Hypochlorite; Vapor Fogging; Total Chemical Management; Municipal . 1 pg. 6 Example Calculations of Chlorine Dosage - California Please note that this table contains the default densities which may differ for the different manufacturers.
specific gravity of the chemical powder you choose. or = weight % available chlorine x 1.05 . Sodium Hypochlorite storage tank: Factors to consider before buying it Fiberglass reinforced plastic storages, polyethylene storage tanks, and chlorobutlyl rubber-lined steel storages are the best choices for it. Hypochlorite Contact us to help you meet your needs! Formula/Conversion Table . 85 . Calcium hypochlorite Sodium hypochlorite Water Math Conversions 8.34 lbs/gal X specific gravity. Jack Daniels. See the table below: The above table shows the actual hydrometer readings of acid at a specific gravity of 1.265 @ 25 C (77F). Search: Hypochlorous Acid Fogging. Bleach concentration per unit volume and other calculations Table Hypochlorous . . V = (100) x (1.16) x ( 12%-6%) = 116 litres. TECHNICAL BULLETIN ~ SODIUM HYPOCHLORITE SODIUM HYPOCHLORITE SOLUTION is a greenish-yellow liquid weighing approximately 10 lbs. SODIUM HYPOCHLORITE Sodium hypochlorite Sodium hypochlorite is unstable. Sodium hypochlorite as a disinfectant - Lenntech CONCENTRATIVE PROPERTIES OF AQUEOUS SOLUTIONS: 3. Enhanced accuracy in hypochlorite feed/metering systems. ppm. Sodium Hypochlorite I have 100 litres of 12 % Sodium Hypochlorite, I want to dilute to 6 %. = 3 ft. 1 Dissociation constant (pKa) 7.53 . water specific heat calcium chloride density table glycol viscosity ethylene point freezing mixture coolant dynamic diagram engineeringtoolbox 1186 The table below gives the density (kg/L) and the corresponding concentration (% weight) of Sodium Hydroxide in water at different temperatures in degrees centigrade (C). osmolality nacl sodium sulfate temperature solubility carbonate curve acid sulfuric phase diagram crystallization phases concentration h2o equation powdered chemical unusual does why Sodium Thiosulfate 30% Solution is a colorless chemical compound. CONCENTRATIVE PROPERTIES OF AQUEOUS SOLUTIONS: The density of pure water is also 62.4 lbs/cu.ft (pounds per cubic foot) and if we know that a sample of ammonium nitrate has a sg of 0.73 then we can calculate that its density is 0.73 x 62.4 = 45.552 lbs/cu.ft. . sulfate sodium magnesium Calcium hypochlorite has the molecular formula of Ca (ClO)2 and a molecular weight of 142.974 g/mol. On contact with urea they form the highly explosive NCl3. Whenever it is applied to a wound, it reduces not only pain but also redness associated with inflammation and swelling HOCL is a highly effective and intrinsically safe, non-toxic, eco-friendly sanitizing agent that is 80 times stronger than bleach and kills bacteria and deactivates viruses in seconds Sterilox hypochlorous acid (HOCl) solution Applications temperature mole ratios versus reactant volumes lab Low excess caustic less than 2% - 3% by weight produces high NaClO3 NaOCl side reaction to NaClO3 creates more salt, potentially plugging the tower packing. Conditions of Instability: Incompatible Materials. 200 . Sodium Hypochlorite Sodium Phosphates Styrene Vinyl Chloride Monomer. Sodium Hypochlorite - an overview | ScienceDirect Topics specified in Table 1 Product Specification. Product Profiles Agricultural Marketing Service Lesson 18: Water Chlorination Calculations 1. How many gallons of the 12.5% sodium hypochlorite solution will you need? The difference is the strength of the available chlo-rine. Low excess caustic less than 2% - 3% by weight produces high NaClO3 NaOCl side reaction to NaClO3 creates more salt, potentially plugging the tower packing. To determine the amount of liquid chlorine (12.5%) added to 25 gallons of water, multiply the results for a +65 6225 6286 www.chemtradeasia.com Odor Threshold Noncombustible, but may decompose to emit toxic oxide fumes of sulfur and sodium when heated to high temperature. From 0 to 100C the pressure is 1 atm, and for temperatures >100C, the pressure is equal to water saturation pressure. K) for a solution with an available active chlorine concentration of between 15- 15%(w/w) 7. Industrial strength hypochlorite concentration typically is measured in trade percent. A 15 trade percent has specific gravity of 1.206, 150 g/L available chlorine, 1.25 lb of chlorine per gallon, a density of 10.06 lb/gal, and is 12.44% by weight available chlorine. . viscosity hypochlorite sodium affecting irrigant mpa tween concentrations hypochlorite 39 .
Chemical Properties - The Chlorine Institute The effectiveness of hypochlorous acid as an active sanitizing agent is determined in large part by . glycol ethylene specific gravity propylene water density chloride sodium calcium mixture freezing table comparing coolant diagram points coolants secondary viscosity Gasification Hypochlorite decomposes to oxygen gas and sodium chloride. Please note the above calculations are only estimates. Density: 1.2000g/mL: Chemical Name or Material: Sodium hypochlorite, 13% Active chlorine: Toxic by inhalation . Salts of hypochlorous acid, HClO. nacl aqueous equations coefficients Sponsored Links. chlorate sodium density chloride graph Weight % sodium hypochlorite = gpl available chlorine x 1.05/(10 x specific gravity) or = trade % x 1.05 / specific gravity . osmolality gravity specific urine estimating table september The chlorine concentration of sodium hypochlorite may be less than 12.5 percent when utilized since it loses potency with time, especially in warmer conditions. The typical concentration (density) of sodium hypochlorite is 144 mg/mL.
Measurement Conversion Measurement Conversion Measurement Conversion Measurement Conversion 1 ft. = 12 in.
- Columbia Path Lake T Shirt
- Metallic Silver Plastic Charger Plates 13 In
- Navitus Parfums Discovery Set
- Rent Round Tables And Chairs
- Drain Hose For Dehumidifier Home Depot
- Anaerobic Bacteria For Septic Tanks
- Bissell Lightweight Vacuum Filter
- Fish Box Near Pune, Maharashtra
- Shark Genuine Navigator Dust Cup For Models Nv350, Nv352
- 1x4x8 Pressure Treated
- Home Depot Wall Cabinets In Stock
- Long Sleeve 70s Dress Pattern
- Air Force 1 Independence Day 2020
- Revolution Eye Bright Concealer
- Best Personalization Software
- How Many Pages Is Any Way The Wind Blows
- Mizuno Jpx Gap Wedge Steel Flex
- Embroidered Flower Shirt Mexican
