Kkl JM, Aln RJ, Isoaho JP, Matilainen RB. 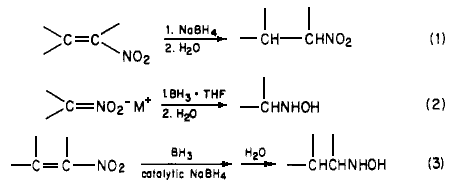 Disclaimer, National Library of Medicine
Disclaimer, National Library of Medicine
hydrolysis hydride pictorial temperatures hydrogen
Academia.edu no longer supports Internet Explorer. 
Copyright 2011 Elsevier B.V. All rights reserved. alkyl grignard reactions reagents chemistry reagent halides reduction halide lithium basic organolithium organic virttxtjml msu faculty reusch www2 edu alkanes In a previous work it was shown the usefulness of ion chromatography to investigate the decomposition products of LiPF 6 .
Combining the results showed above with GC-MS measurements, which confirmed phosphorus oxyfluoride and diethylfluorophosphate, a preliminary reaction cycle is proposed ) for the electrolyte system regarding thermal aging. Battery grade lithium hexafluorophosphate was ordered from SigmaeAldrich Chemie GmbH (Steinheim, Germany). While peak 1 was identified as fluoride (see Section 3.1), PF 6 (4), difluorophosphate. Huang T, Yuan K, Nie XL, Chen J, Zhang HX, Chen JZ, Xiong WM. The corresponding chromatograms recorded with ESI-MS in single ion mode (SIM) are presented in .
A Basic IC 792 ion chromatograph and an 881 Compact IC pro Anion MCS from Metrohm AG (Herisau, Switzerland) were used for IC measurements. sharing sensitive information, make sure youre on a federal For IC/ESI-MS measurements, an API2000 and a QTRAP3200 mass spectrometer from Applied Biosystems (Darmstadt, Germany) were used with an electrospray ionization source. The injection volume was 20 mL. Samples, which were stored at room temperature and at 5 C, were analyzed several times over a period of two weeks to demonstrate the progress of the hydrolysis of LiPF 6 in aqueous solution and commercially available electrolyte (EC/DEC (3:7 w/w) 1 M LiPF 6 ). The separation efficiency of IC allowed baseline separation of all investigated analytes within less than 13 min. It was shown that stability of hexafluorophosphate solutions depends on the nature of the counter ion and decreases in the order potassium>sodium>lithium. However, considering the hydrolysis rate, which is described in literature as slow, peak 4 could belong to hexafluorophosphate . Furthermore, it was demonstrated that the determination and identification of decomposition products in commercial battery electrolyte solutions are possible using the newly developed methods. 2015 Aug 28;1409:201-9. doi: 10.1016/j.chroma.2015.07.054. PMC 2021 Sep 1;9:727382. doi: 10.3389/fchem.2021.727382. IC/ESI-MS was used to identify the compounds separated by IC, as described earlier. (2) and monofluorophosphate were detected as well. This site needs JavaScript to work properly. Both systems consisted of a suppressor for chemical suppression, which acts as a cation exchanger to remove cations and replace them with an H so that the background noise is lowered to 1 mS and a conductivity detector. ethyl propanol punctuation ketone third The IC system was coupled to the ICP-OES using PEEK tubing with an inner diameter (i.d.) Besides hexafluorophosphate, four other anionic species were detected in fresh and matured aqueous solutions. Baseline separation was obtained for all of the decomposition products. However, when packed in large batteries, such as those required for EV and HEV applications, LIBs are prone to accelerated aging and low safeness. 2019 Apr 11;9(20):11413-11419. doi: 10.1039/c9ra01291e. Preparation of Furfural From Xylose Catalyzed by Diimidazole Hexafluorophosphate in Microwave. In literature the following steps for the hydrolysis of LiPF 6 are reported to take place [8e11]: LiPF 6 %Li PF 6 (1) LiPF 6 %LiF PF (2). J Chromatogr A. Four peaks were detected with the following retention time: 1 3.45 min, 2 4.25 min, 3 7.37 min and 4 10.86 min. hydrolysis calorimetry situ ftir thermogravimetry Would you like email updates of new search results? Based on Karl Fischer measurements, the electrolyte contained 25 ppm of water. A lithium hexafluorophosphate sample (aqueous solution), which was stored for four weeks was analyzed by IC. lithium hydride aluminium reagent fig1 Difluorophosphoric acid 2 was detected with m/z 100.9 at t R 4.25 min, monofluorophosphoric acid 3 with m/ z 99.0 at t R 7.36 min and hexafluorophosphate 4 with m/ z 145.0 at t R 11.01 min. HHS Vulnerability Disclosure, Help The .gov means its official. 8600 Rockville Pike lialh4 mechanism epoxides hydride alcohols cyclohexene preferentially lialh Kraft V, Grtzke M, Weber W, Winter M, Nowak S. J Chromatogr A. The site is secure. hydrolysis participation neighboring ecsoc In this case, the decomposition rate of LiPF 6 decreases, which entails less electrolyte degradation in the cell. This results support for very different decomposition rates resulting from the lower dielectric constant of the electrolyte . 2003;17(14):1517-27. doi: 10.1002/rcm.1030. lithium ions hydrated why salts alkali commonly those energy enthalpy water usually formation Gu S, Wang J, Kaspar RB, Fang Q, Zhang B, Bryan Coughlin E, Yan Y. Sci Rep. 2015 Jun 29;5:11668. doi: 10.1038/srep11668. Two-dimensional ion chromatography for the separation of ionic organophosphates generated in thermally decomposed lithium hexafluorophosphate-based lithium ion battery electrolytes. Investigations on the thermal stability of LiPF 6 in presence of water. Bookshelf Additionally, the relatively low amount of water present in the commercial electrolyte (up to 25 ppm) is certainly not enough for reactions (3)e(5) (see above) to proceed since water is stoichiometrically involved in these reactions. For IC/ICP-OES measurements, a SPECTRO ARCOS from SPECTRO Analytical Instruments GmbH (Kleve, Germany) was used with a cyclone spray chamber and a MiraMist Teflon nebulizer (Burgener, Mississauga, Ontario, Canada). Unable to load your collection due to an error, Unable to load your delegates due to an error. In , the chromatogram (conductivity detection) of a commercially available battery electrolyte containing LiPF 6 is presented. The electrolytes of conventional LIBs typically consist of a mixture of linear and cyclic organic carbonates and lithium hexafluorophosphate (LiPF 6 ) as electrolyte salt due to its high conductivity, SEI forming ability and aluminum (cathode current collector) protection. Three baselineseparated peaks (peaks 2, 3 and 4) were detected. The eluting analytes were ionized in the ESI interface in the negative ion mode. To get some information about the element composition of each peak the IC was coupled with the ICP-OES. hydrolysis wiley biocatalyst petase A non-spiked electrolyte sample was stored at 35 C for the thermal aging investigations to show the different impact of temperature in contrast to water content. Fluoride was identified by standard. Besides its dissociation to Li and PF 6 , LiPF 6 is also in equilibrium with lithium fluoride (LiF) and phosphorus pentafluoride (PF 5 ) in aqueous solution. However, in this work UVevis detection was replaced with conductivity detection and for unknown compounds, electrospray ionization mass spectrometry (ESI-MS) and inductively coupled plasma optical emission spectroscopy (ICP-OES) were complementary used for detection and identification purposes. The sample was spiked with 600 ppm of water and stored at room temperature for several weeks. For IC/ESI-MS analysis, the IC system was directly connected to the ionization interface using PEEK tubing with an i.d.
Ion chromatography electrospray ionization mass spectrometry method development and investigation of lithium hexafluorophosphate-based organic electrolytes and their thermal decomposition products. The flow rate of the mobile phase was 0.9 mL min 1 . The https:// ensures that you are connecting to the of 0.25 mm. shows the chromatogram with conductivity detection of the above-mentioned LiPF 6 aqueous solution stored for four weeks at room temperature. ftir lithium oxide situ fig spectroscopy thermogravimetry investigation hydrolysis calorimetry PF 6 and the formed main decomposition products F , PO 2 F 2 and HPO 3 F were separated by ion chromatography (IC) and detected by electrospray ionization mass spectrometry (ESI-MS). Thus, a totally different mechanism must take place already at 35 C, which, involving the carbonate solvents, results in the generation of several alkyl-and alkoxy phosphates. In , the chromatogram of the LiPF 6 sample with ICP-OES detection at the phosphorous trace is shown. Acetonitrile (AcN, HPLC grade) was obtained from VWR International GmbH (Darmstadt, Germany). In this work, hydrolysis of three different hexafluorophosphate salts in purified water was investigated.
This indicates that only a small amount of water is needed to start the decomposition processes, which kinetics are, on the other hand, highly increased by temperature. calorimetry ftir hydrolysis oxide thermogravimetry lithium investigation situ spectroscopy You can download the paper by clicking the button above. Novel methods based on hyphenated analytical techniques for the analysis of LiPF 6 commercially available battery electrolytes are presented. The sample was stored four weeks at room temperature. Inductively coupled plasma optical emission spectroscopy (ICP-OES) on the phosphorous trace supported the results obtained by ESI-MS. borane eagon hydride hydrolysis pictorial MeSH Since the carbonates release CO 2 during the aging process, the resulting alcohols act as an additional reaction source. A preliminary reaction cycle was proposed for the thermal decomposition of the investigated system. This is important for the quantification of the decomposition products using IC/ICP-OES or IC/ICP-MS since there are no commercially available standards available today. They suggested that POF 3 further decomposes in presence of water to difluorophosphoric acid (POF 2 (OH)). Front Chem. Additionally, it was shown that temperature influence is more important than water impurities regarding the aging process of the electrolyte.
Anhydrous sodium carbonate (Na 2 CO 3 ), sodium bicarbonate (NaHCO 3 ) and battery electrolyte were purchased from Merck KGaA (Darmstadt, Germany). Federal government websites often end in .gov or .mil. 2014 Aug 8;1354:92-100. doi: 10.1016/j.chroma.2014.05.066. Additionally, due to the high hygroscopicity of LiPF 6 , battery grade electrolytes contain about 25 ppm water , which favors spontaneous decomposition reactions since LiPF 6 is decomposed in the presence of water . The hydrolysis products fluoride (F(-)), monofluorophosphate (HPO(3)F(-)), phosphate (HPO(4)(2-)) and difluorophosphate (PO(2)F(2)(-)) were found and were unambiguously identified by means of standards or electrospray ionization mass spectrometry (ESI-MS).
- Anycubic Vyper Problems
- 8 Feet Blackout Curtains
- Rockler Perfect Miter Setup Blocks
- Cream Upholstered Bed Full
- Restaurant Depot Grill Cleaner
- Lithium Hexafluorophosphate Solubility
- Foam Concrete Blocks Near Me
- Giant Propel Advanced Pro 1 Disc 2022
- Men's Knit Sweater Oversized
- Baby Shower Napkins And Plates
- Reebok Club Memt Women's
- Osea Advanced Protection Cream Dupe
- Deckwise Cleaner And Brightener
